Research
Alveolar dysfunction associated to airway allergy
Respiratory disorders caused by allergic reactions have been associated to bronchiolar inflammation leading to bronchoconstriction and life-threatening narrowing of airway lumen. However, whether airway allergy causes alveolar dysfunction compromising respiratory function and, eventually, contributing to asthma remains unaddressed.
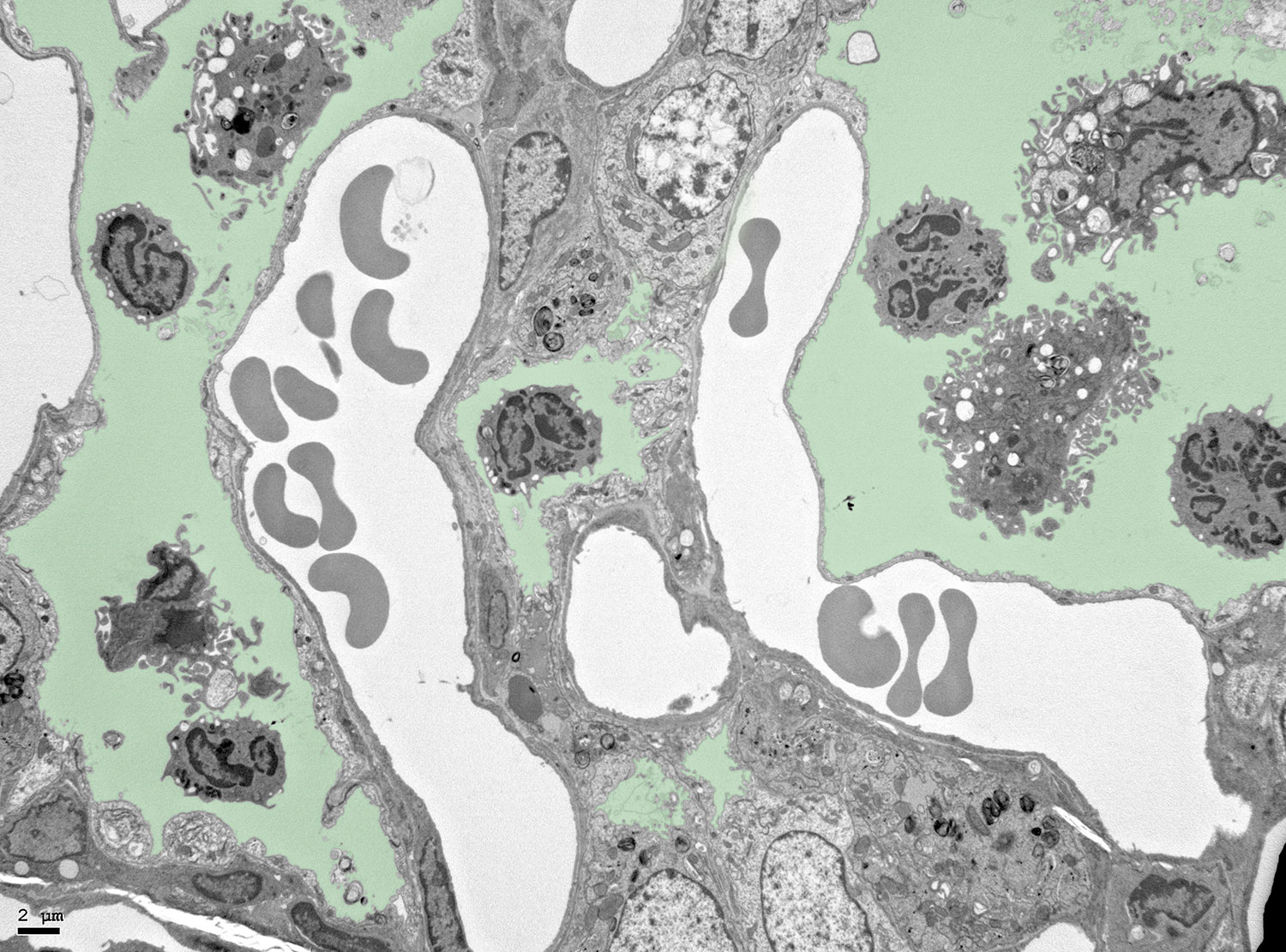
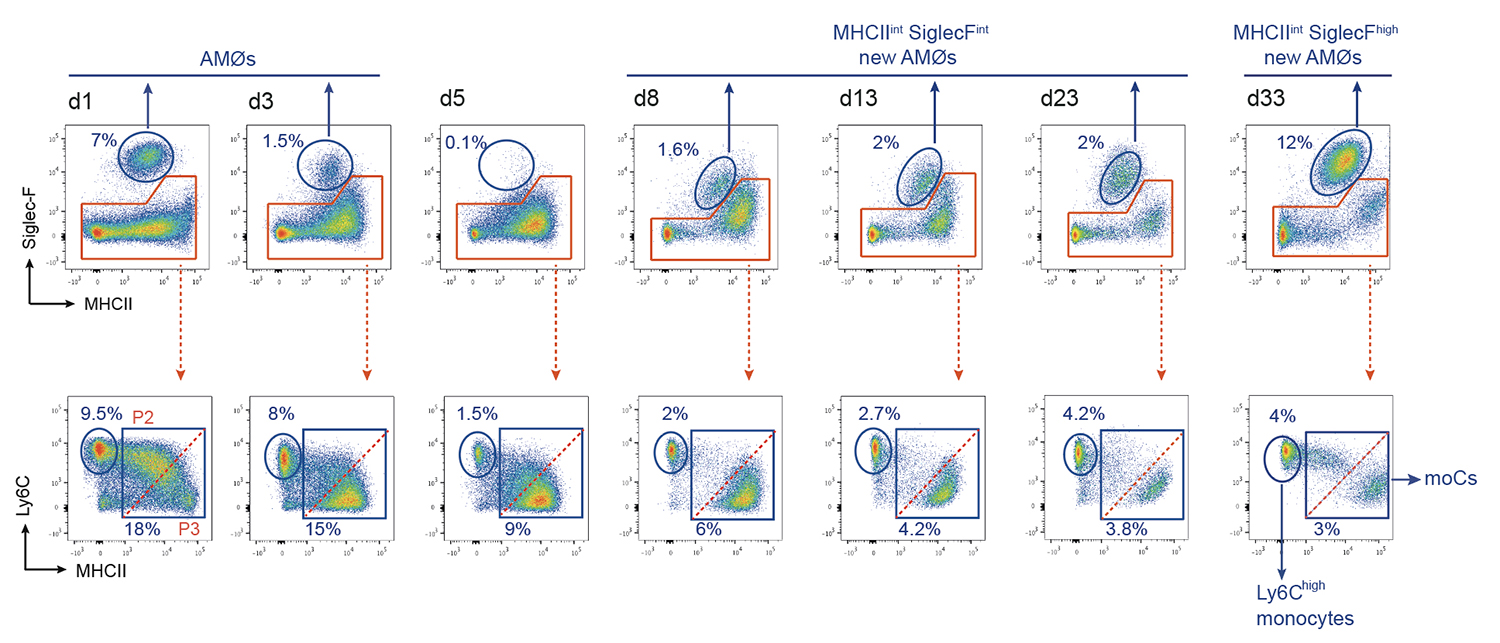
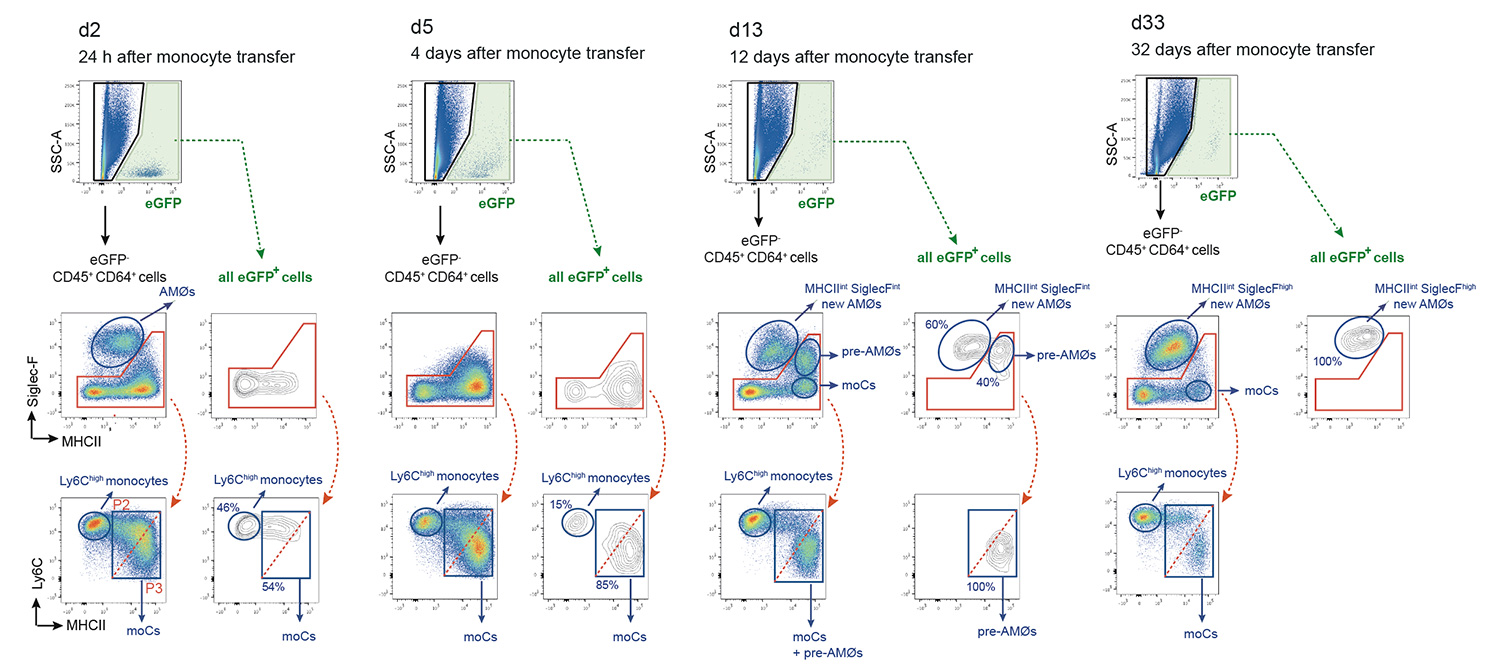
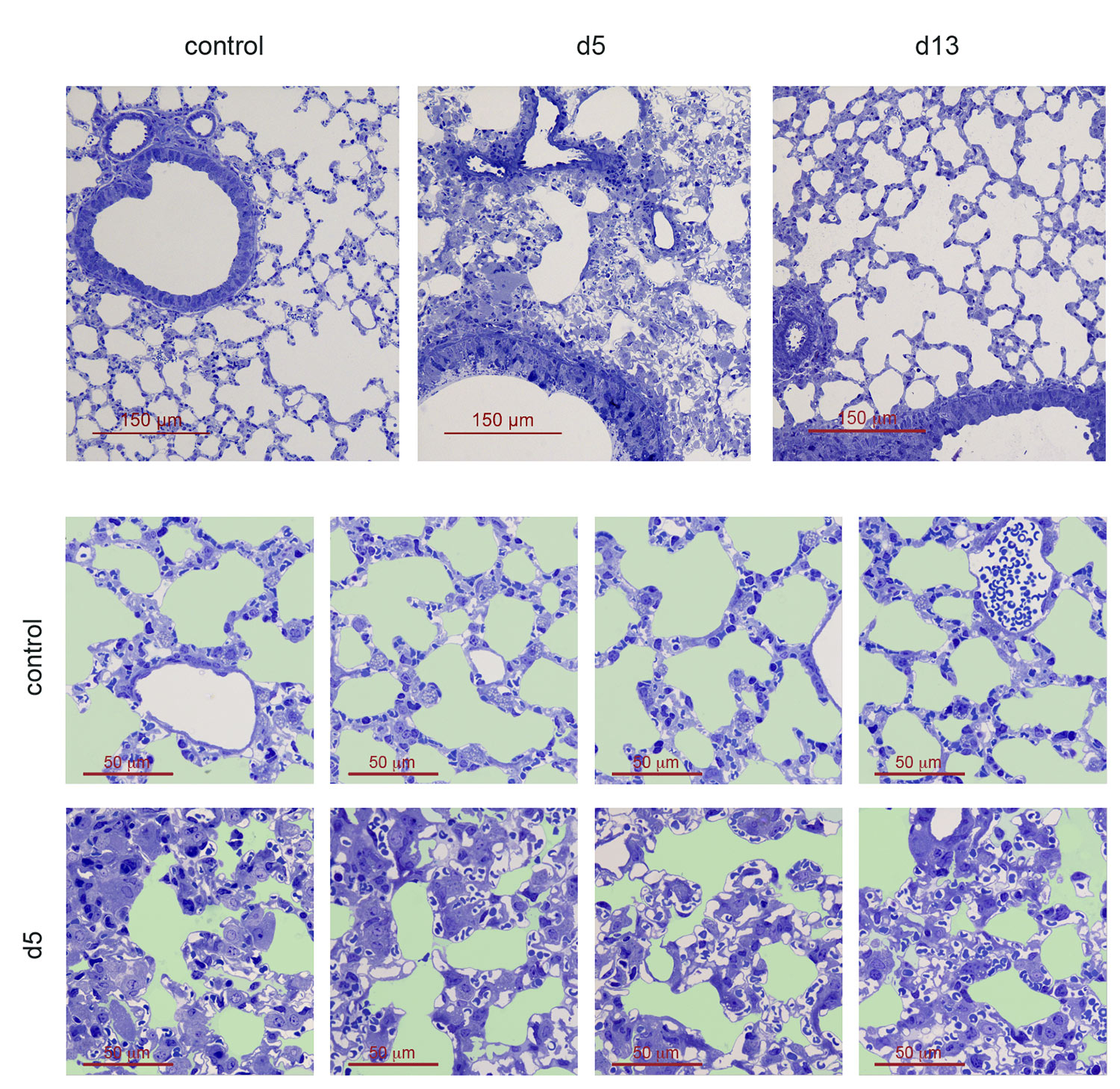
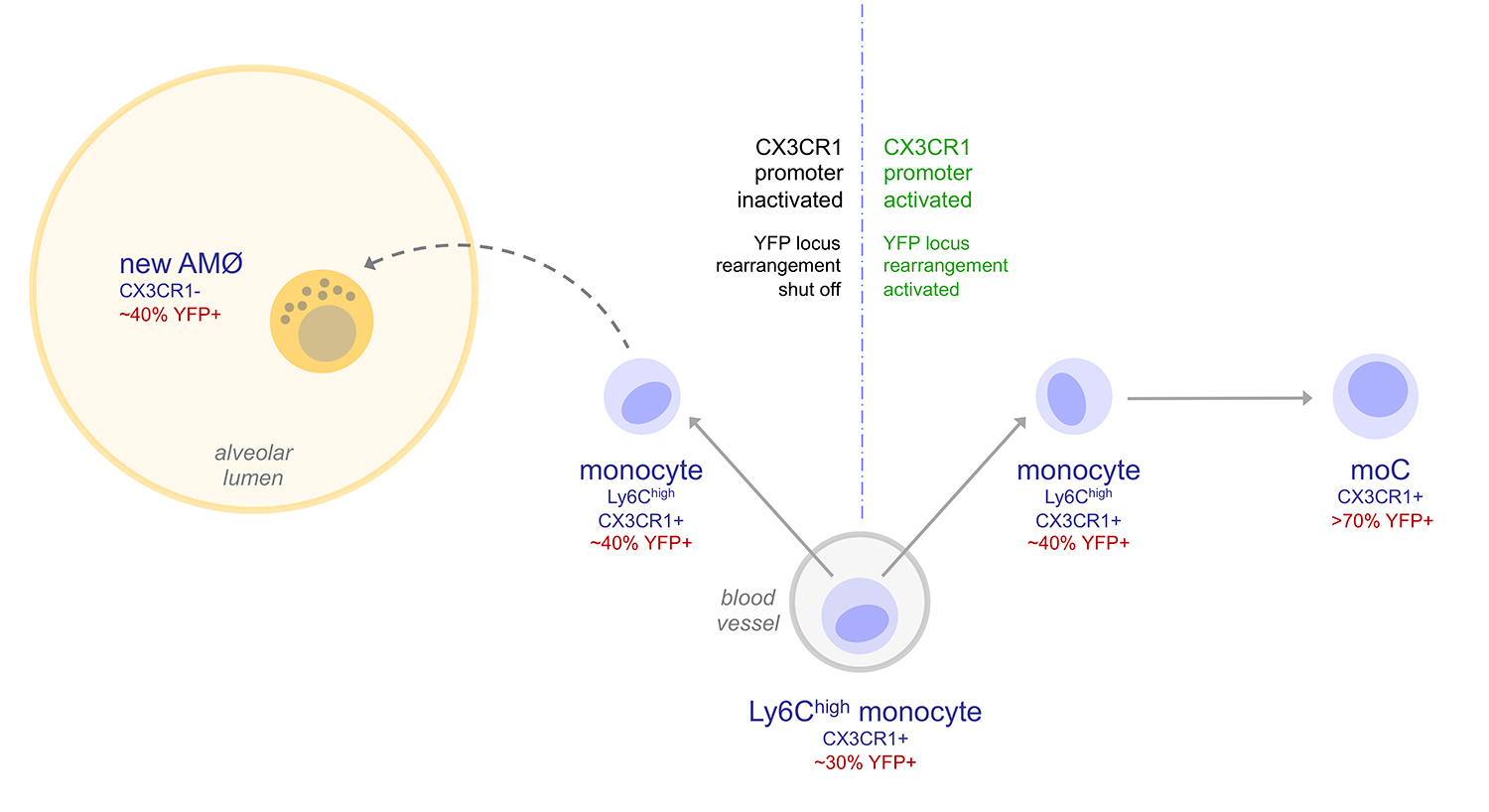
Using a mouse model of house dust mite (HDM)-induced airway allergy, our results revealed that HDM allergy causes severe alveolar disorganization, involving the disappearance of alveolar macrophages (AMØs), pneumocyte hypertrophy, thickening of the alveolar lining and surfactant dysfunction. SP-B/C surfactant proteins are reduced in allergic lung surfactant, that displays a decreased efficiency to form surface-active films able to maintain low surface tensions, as assessed by captive bubble surfactometry, increasing the risk of atelectasis. The analysis of alveolar macrophage regeneration in Cx3cr1cre:R26-yfp chimeras demonstrated that original AMØs are replaced by a new population of monocyte-derived AMØs, that persist long after the resolution of allergy. Monocyte to AMØs transition occurs through an intermediate pre-AMØ stage and is concomittant with translocation into the alveolar space, Siglec-F upregulation and CX3CR1 downregulation (Feo-Lucas et al., Frontiers in Immunology 2023)
These data support that the severe respiratory disorders caused by asthmatic reactions not only result from airway inflammation, but also from profound alterations in the alveolar system, supporting that new therapeutic strategies against asthma shoud be designed based a combined treatment of bronchiolar inflammation and alveolar dysfunction.
Current research in Carlos Ardavin lab in the context of allergy-associated alveolar dysfunction is focused on exploring the molecular mechanistic basis of AMØs death and pneumocyte hypertrophy occurring during the peak of the airway allergic reaction.